
Understanding IEC 60601-1-2:2024 EMC Requirements
A comprehensive guide to the latest electromagnetic compatibility standard updates and their impact on medical device design.
MedMI provides a pre-qualified, reusable HMI and compute platform that eliminates certification bottlenecks and accelerates time-to-market for medical device manufacturers.

Faster Certification
vs. custom HMI development
Across the medical device industry, HMI hardware is repeatedly designed, integrated, and certified for each product—despite offering minimal differentiation.
of device BOM concentrated in HMI subsystems, making it a significant cost driver
IEC 60601 / EMC testing repeated for every SKU, regardless of similarities
average certification timeline per product, delaying market entry
Create a centrally qualified HMI subsystem that OEMs integrate as a proven component—dramatically reducing redesign and recertification effort.

Plug & Play
Ready to integrate
Our platform is qualified to the essential medical device standards, reducing your certification burden and accelerating market entry.
Safety and essential performance requirements for medical electrical equipment
EMC immunity and emissions compliance for medical devices
Application of usability engineering to medical devices
Software lifecycle processes with segregation templates
Aligned with FDA cybersecurity expectations and requirements
General Safety and Performance Requirements for EU market
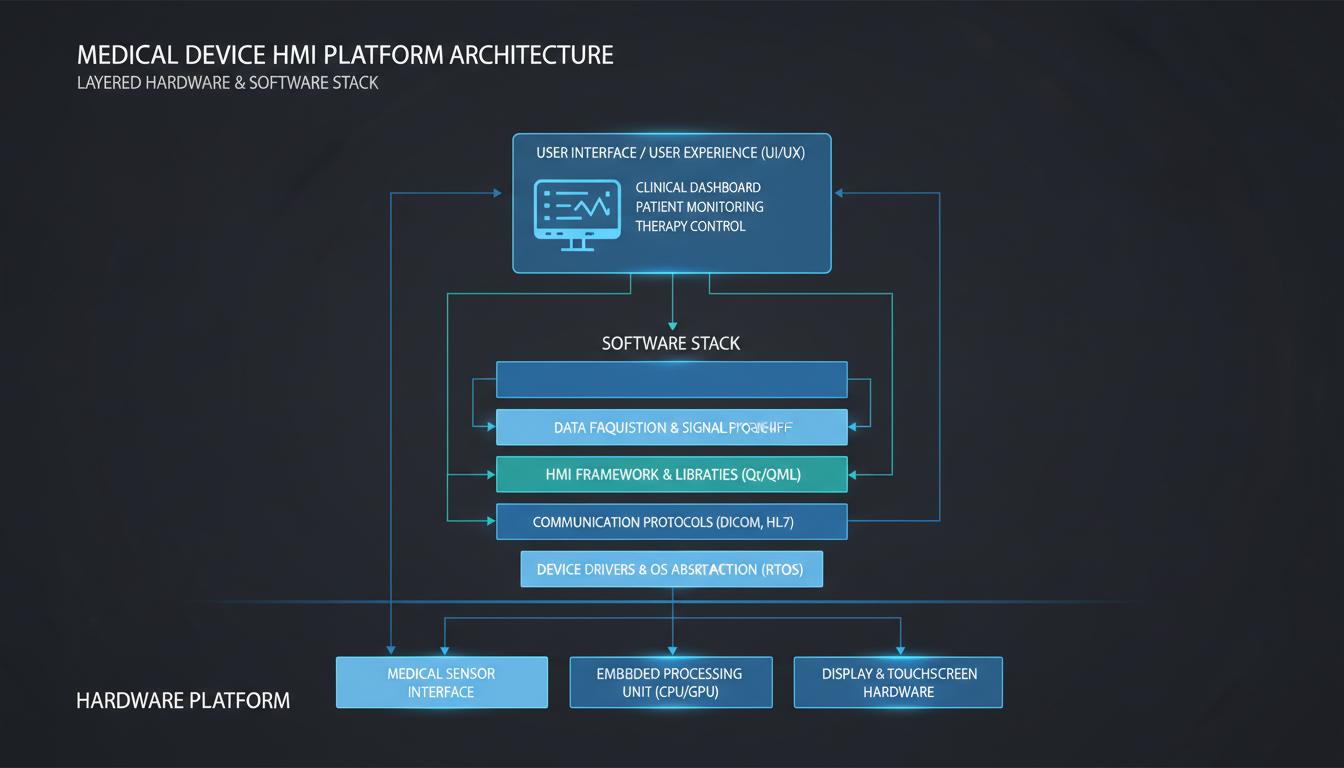
MedMI delivers measurable improvements across cost, time, and risk dimensions for medical device manufacturers.
Aggregated volumes across OEM ecosystem deliver significant component cost reductions
Eliminate repeated certification cycles and accelerate product launches
Proven, qualified platform reduces regulatory rework and certification risk
Benefit from collective purchasing power and long-term supply agreements
Create reusable internal platform that compounds value across product lines
Once embedded in DHFs, creates long-term defensibility and customer retention

A comprehensive guide to the latest electromagnetic compatibility standard updates and their impact on medical device design.
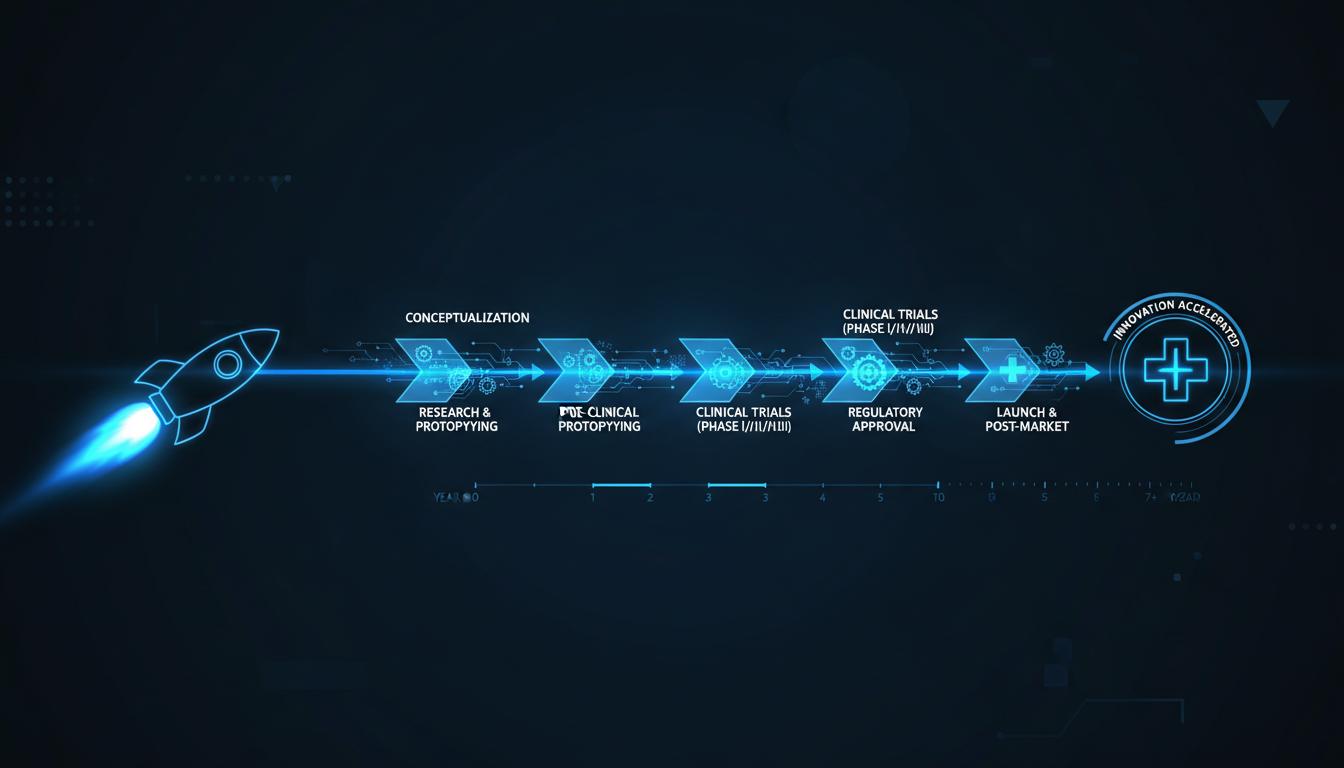
How medical device OEMs are leveraging qualified subsystems to accelerate certification and reduce regulatory risk.
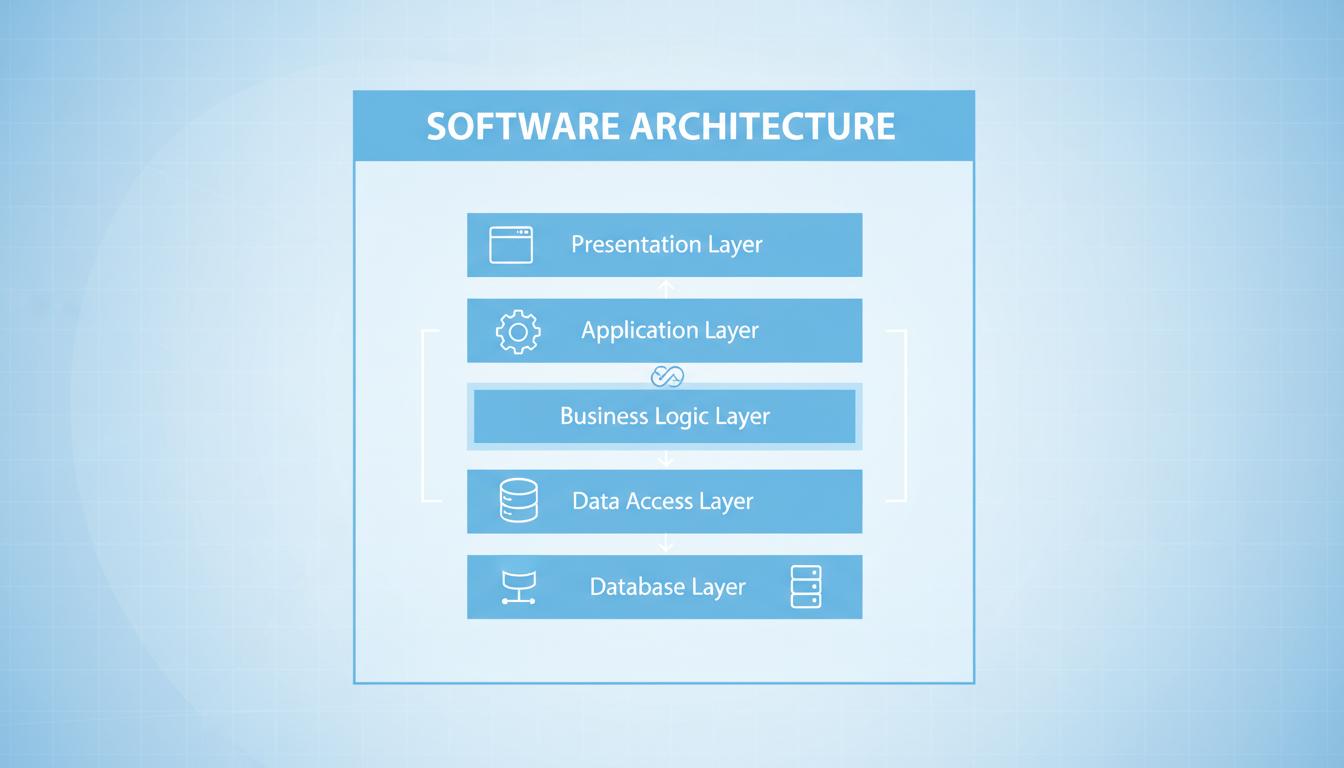
Implementing effective software segregation strategies to meet safety classification requirements.
Analysis of cost structures and savings opportunities from platform-based HMI development approaches.

Addressing FDA and MDR cybersecurity requirements in modern medical device interfaces.

How regulatory bodies view pre-qualified subsystems and platform-based device development.
Schedule a consultation with our technical team to discuss how MedMI can reduce your certification timeline and BOM costs.